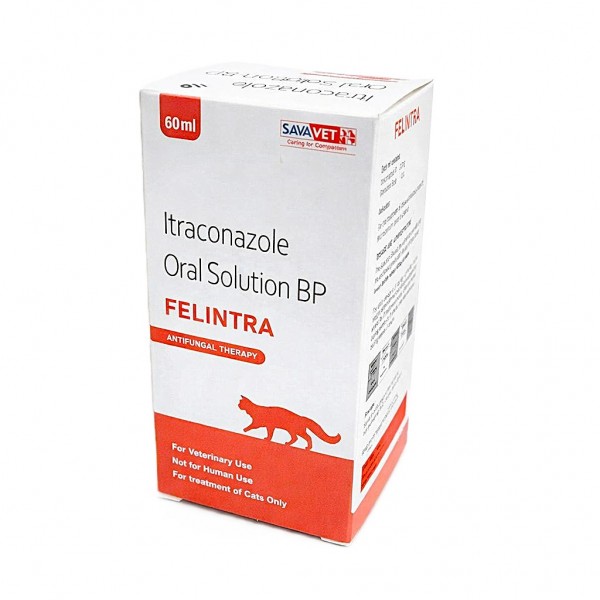
FELINTRA Itraconazole Oral Solution 10mg/ml for Cats 60ml – Antifungal Treatment for Ringworm (Microsporum canis) | Veterinary Medicine with Dosing Syringe for Feline Skin Infections
-600x600w.jpeg)
-600x600.jpeg)
-600x600.jpeg)
-600x600.jpeg)
-80x80w.jpeg)
-80x80.jpeg)
-80x80.jpeg)

-80x80.jpeg)
- حالة التوفر: متوفر
- رمز الموديل: VL8905045002540
FELINTRA Itraconazole Oral Solution BP is a premium veterinary antifungal medicine specially formulated for cats to treat dermatophytosis (ringworm), particularly infections caused by Microsporum canis. This advanced oral solution contains itraconazole 10 mg/mL, a clinically proven antifungal agent that effectively eliminates fungal infections while supporting healthy skin and coat recovery.
Designed for easy and accurate administration, FELINTRA comes with a graduated dosing syringe, ensuring stress-free dosing for both pets and pet owners. Its highly bioavailable formula allows rapid absorption and deep penetration into skin and hair tissues, delivering long-lasting antifungal action.
Composition
Each 1 mL contains:
- Itraconazole IP – 10 mg
- Flavoured base – q.s.
Indications
- Treatment of dermatophytosis (ringworm) in cats
- Effective against Microsporum canis
- Helps restore healthy skin and fur
- Controls spread of fungal infections
Mechanism of Action
Itraconazole belongs to the triazole antifungal class. It works by selectively inhibiting fungal cytochrome P450 enzymes, which are essential for ergosterol synthesis. Ergosterol is a key component of fungal cell membranes. By blocking its production, FELINTRA causes irreversible damage to fungal cells, ultimately leading to their death.
Clinical Pharmacology
Pharmacodynamics
- Strong antifungal activity against dermatophytes
- Targets fungal membrane integrity
- Causes structural degeneration of fungi
Pharmacokinetics
- Rapid absorption after oral administration
- Higher absorption when given with food
- Plasma protein binding greater than 99%
- Widely distributed in tissues, especially skin and hair
- Peak concentration reached in approximately 2 hours
- Half-life increases with repeated dosing (up to ~36 hours)
- Eliminated primarily through feces
Itraconazole accumulates in hair during treatment, ensuring prolonged antifungal protection even after dosing stops.
Dosage & Administration
- Recommended Dose: 5 mg/kg body weight (0.5 mL/kg)
- Route: Oral (use provided dosing syringe)
Treatment Schedule (Pulse Therapy)
- Week 1: Daily dosing (7 days)
- Week 2: No treatment
- Week 3: Daily dosing (7 days)
- Week 4: No treatment
- Week 5: Daily dosing (7 days)
This alternating-week schedule improves treatment success while reducing the risk of drug accumulation.
Packaging
- 60 mL bottle
- Includes graduated dosing syringe for precise measurement
Contraindications
- Do not use in cats with known hypersensitivity to itraconazole
Warnings & Precautions
- Not proven safe for pregnant or lactating cats
- Use only under veterinary supervision
- Prescription may be required
- For animal use only – not for human use
Possible Side Effects
- Mild gastrointestinal disturbances such as vomiting or reduced appetite
- If symptoms persist, consult a veterinarian
Drug Interactions
Use caution when combining with:
- Macrolide antibiotics (erythromycin, clarithromycin)
- Antacids and proton pump inhibitors (omeprazole)
- Phenobarbital, phenytoin, rifampin
- Digoxin, warfarin
- Benzodiazepines (diazepam, midazolam)
- Corticosteroids
- Cyclosporine, ivermectin
These medications may alter itraconazole levels or increase risk of side effects.
Additional Scientific Information
Itraconazole is a racemic mixture consisting of four diastereomers, each contributing to its antifungal activity. It binds strongly to fungal enzymes and remains active in keratin-rich tissues such as skin and hair, making it highly effective for dermatological infections in cats.
Storage Instructions
- Store below 25°C
- Keep in a dry place
- Protect from light
- Keep out of reach of children
Manufacturer & Marketing Details
- Manufactured by: Akums Drugs & Pharmaceuticals Ltd., Uttarakhand, India
- Marketed by: Sava Healthcare Limited (Savavet), Gujarat, India
Product Information
- Net Quantity: 60 mL
- Batch No.: PKAB08
- Manufacturing Date: December 2024
- Expiry Date: December 2026
- Mfg. License No.: 123/UA/2007
Important Notice
- For veterinary use only
- Not for human use
- Use only for cats
- To be administered under guidance of a registered veterinary practitioner
- Read package insert carefully before use
Category
Veterinary Medicines → Cat Medicines → Antifungal Treatments






-350x350h.jpeg)
-350x350w.png)
-350x350.jpeg)